_Kara Spiller
Spiller is an assistant professor in the School of Biomedical Engineering, Science and Health Systems.
Scientists are getting closer to mending damaged tissue and even creating whole organs, thanks to advancements in regenerative medicine. But a significant roadblock exists: the absence of blood vessels inside damaged tissues.
Spiller has been studying ways to naturally spawn blood vessels to feed new tissue growth by taking advantage of the body’s response to injury and disease. She believes that vital immune system cells, called macrophages, are key to that process.
“Damaged tissue — due to cancer, a bone fracture or a disease like diabetes — happens because insufficient blood vessel growth has cut off oxygen. If we cannot engineer blood vessel networks, then we cannot repair any tissue.”
The National Institutes of Health has awarded her with a five-year, $1.9 million grant to study the cells.
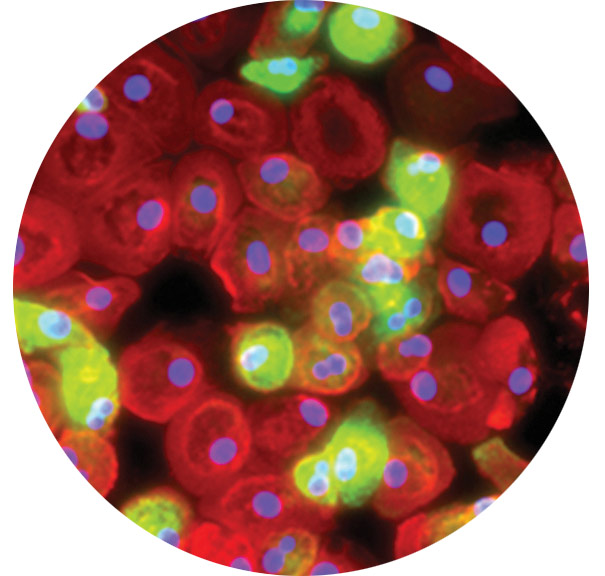
Macrophages roam throughout tissues in the body and engulf any pathogens in their path. Though these immune cells destroy foreign invaders, they have earned a bad reputation, Spiller says, because they also contribute to inflammation, causing bruises, ulcers and sometimes, degenerative diseases.
A microscopic view of macrophages, immune system cells that may be key to blood vessel growth.
However, more recent studies have shown that macrophages, and inflammation more generally, can play an important role in tissue repair and regeneration, due to the cells’ ability to change their form and function — each orchestrating events specific to different stages of repair. Though this process has been identified, it is not well understood.
Once macrophages are better understood, Spiller and her research team hope to develop a biomaterial platform and strategy for delivering drugs to control macrophages and encourage vascularization by the body’s own cells.
“The idea is that if you can manipulate the macrophages via just a single drug, then the macrophages will be able to control the blood vessels,” Spiller says.