_MinJun Kim
Kim is an assistant professor of mechanical engineering and mechanics. His focuses are on micro/nanoscale fluid mechanics, biological transport phenomena and more.
Many children of the ’80s remember “Innerspace,” the Steven Spielberg sci-fi comedy starring Dennis Quaid as a man miniaturized inside a tiny submersible during a secret experiment and injected into a human body.
Not everyone who saw the movie thought tiny machines moving through the body were possible.
MinJun Kim did. And he’s trying to make it happen.
“It was a science fiction movie, but it didn’t seem out of reach to me,” jokes Kim, who is a professor of mechanical engineering and mechanics and director of the Biological Actuation Sensing & Transport Laboratory (BASTLab), one of only a small handful of micro and nanorobot research groups in the United States.
Kim has spent his career combining microorganisms with engineering controls to create novel types of biomicrotechnology. As a doctoral student and now as the director of the BASTLab, Kim has tested ways to co-opt the movements of bacteria and protozoa — and later inorganic, biodegradeable beads that move like flagellated bacteria — to get them to perform tasks inside a fluid environment.
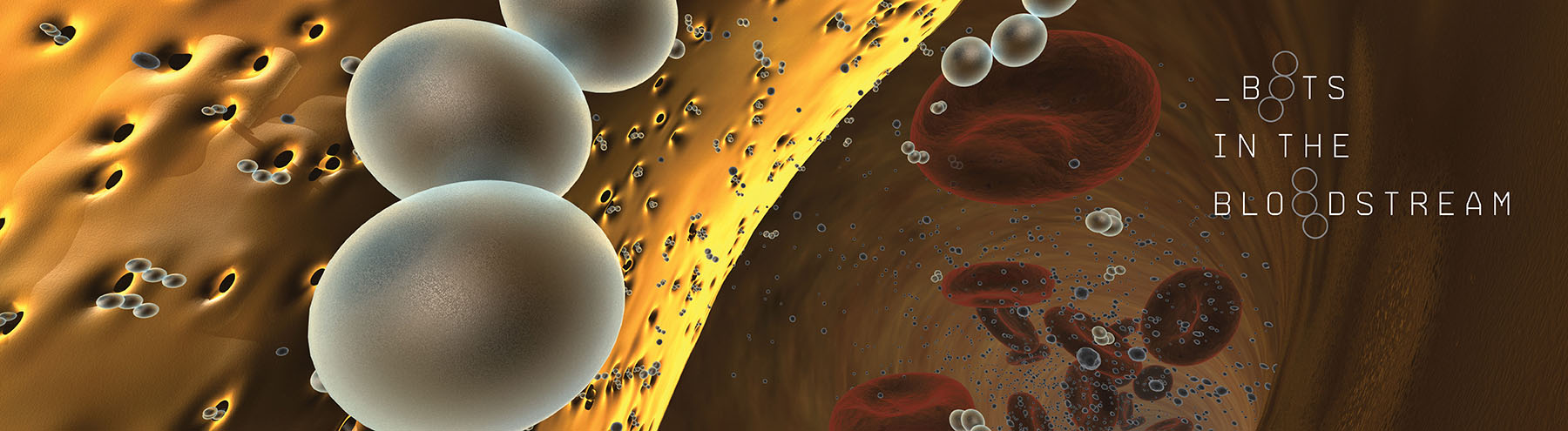
Now, he’s putting his bio-inspired micro and nanorobotics knowledge to work as a member of a new, ambitious international research initiative that aims to create a minimally invasive nanorobotic toolkit for clearing fully blocked arteries within five years.
“Microrobots are scalable, easily manipulated and non-disruptive to the human body; they can do many things, such as deliver medication to tumors or unclog arteries, without using open surgery.”
The $18 million project combines 11 institutions that are home to some of the top roboticists and engineers in the world, including ETH Zürich, Seoul National University, Hanyang University and Samsung Medical Center, among others. The project is backed by the Korea Evaluation Institute of Industrial Technologies and organized by the Daegu Gyeongbuk Institute of Sciences and Technology, a government-funded research entity in South Korea.
Kim’s team, which includes Drexel post-doctoral researcher U Kei Cheang, is the only member of the group from the United States. Kim is already well on his way to having a working model and will test the technology in animal models as early as this summer.
“Microrobotics is still a rather nascent field of study, and very much in its infancy when it comes to medical applications,” Kim says. “Microrobots are scalable, easily manipulated and non-disruptive to the human body; they can do many things, such as deliver medication to tumors or unclog arteries, without using open surgery. A project like this, because it is supported by leading institutions and has such a challenging goal, is an opportunity to push medicine and microbotics into a new and exciting place.”
Beads and magnets
This is only the first year of a five-year partnership, but Kim has been refining the science of his design for a long time already.
Kim’s microrobots, which he calls “microswimmers,” are chains of three or more flexible iron-oxide particles, rigidly linked together through chemical bonds and magnetic force.
In a series of papers published in Physical Review, Applied Physics Letters and the Journal of Nanoparticle Research in 2014 and 2015, Kim and his research team chronicle their progress in achieving control over the beads. In lab tests, using an external electromagnetic field to actuate and control the microswimmers, they’ve been able to combine beads into chains and disperse them at will. They’ve also propelled two different microswimmers in opposite directions simultaneously in the same fluid environment. And they’ve moved the pair at the same and at different speeds, too.

Each microswimmer consists of three beads made of magnetic iron-oxide. To bind the beads strongly to each other, some are coated with the small molecule biotin and others with the bacterial protein streptavidin. The biotin-streptavidin bond is one of the strongest noncovalent interactions in nature.
“We can control speed, direction, orientation, docking and undocking,” Kim says. “It was truly a challenge to develop a 2D/3D magnetic field control system with micro and nanoscale precision as well as control theories and algorithms for in vivo environments.”
The beads are so small — 4.40 micrometers in diameter, about half the size of a red blood cell and a third of the size of a white blood cell — that they can glide easily through the bloodstream.
The beads are put in motion by an external magnetic field that causes each bead to rotate. Because they are linked together, their individual rotations propel them forward in a twisting corkscrew pattern, ideal for burrowing through arterial plaque to loosen blockages in the bloodstream.
The team’s plan is to use the microswimmers in concert with a probe that resembles a tiny drill, which is being designed by another member of the team, Professor of Robotics and Intelligent Systems Bradley Nelson at ETH Zürich. A catheter will be used to deliver the microswimmers and the drill directly to the blocked artery. From there, the swimmers would push their way into the blockage, after which the drill would clear it completely.
The Daegu Gyeongbuk Institute asked Kim to be involved in the project because the organizers believed he might be able to help them achieve better control over the drill robot, Kim says.
“But they would also like to see if our beads can do the drilling,” he says. “Our tool is easier to fabricate; theirs requires at least a week to manufacture.”
As Kim and the team envision it, a trained surgeon will control the microswimmers’ speed and direction inside a patient by manipulating an external magnetic device made up of electromagnetic coils, similar to a mini-MRI.
Kim has engineered a couple of these devices, each about the size of a tissue box, to steer and manipulate micro and nanorobot prototypes in his lab. During the next couple of years, other researchers in the partnership will develop a sophisticated version of the device that doctors can use to direct surgeries. The partnership anticipates testing the technology in lab and clinical settings within in the next four years.
Kim modeled the corkscrew movements of his microswimmers after a bacterium that wreaks havoc in the body by burrowing through healthy tissue. Borrelia burgdorferi, the bacteria that causes Lyme disease, is classified by its spiral shape, which enables its movement. Kim hopes to harness the malicious structure and motion that makes Lyme so destructive to improve lives instead.
“This international collaboration is a way to grab onto the opportunity to introduce our robots to the world,” says Kim. “If our microrobots can drill through clogged arteries, it would help many people who suffer from atherosclerosis.”
Medicine inspired by pathogens
Bacteria — their movements, their shapes — have been at the root of Kim’s robotics since he was a doctoral student at Brown University.
At Brown, Kim experimented with a strain of E. coli bacteria that had been genetically modified by Howard Berg, a Harvard University physics professor (Kim would later become a postdoctoral fellow at the Rowland Institute at Harvard) to have a varying number of flagella. Flagella are lash-like appendages that propel certain microorganisms; by altering their number, the organism’s rotation speed can be controlled by external factors, such as chemicals, specific wavelengths of light and electric fields.
Kim’s idea was to harness E. coli’s motility as a sort of motor to power other things, and in 2006, he attached the bacteria to the bottom of a microfabricated structure to create a bacteria-powered microrobot. He successfully showed that the hybrid could be used for micro-manipulation and obstacle avoidance.
That same year, Kim came to Drexel, where his interests shifted toward applications that could improve human health, one of the main tenets of Drexel’s translational research mission. And while his experiments had shown promise of having some practical applications, E. coli is still E. coli. Even modified to reduce its innate toxicity, E. coli has the potential to startle the immune system and throw the body into chaos. “People don’t want bacteria inside them,” Kim says simply.
Kim continued to dabble in experiments involving artificially magnetotactic (prone to self-align with the Earth’s magnetic field) protozoa until 2011, but by then his team had largely switched their focus to man-made biodegradeable particles, such as the microswimmers he is currently studying as a treatment for blocked arteries.
Not only do non-cell-based materials eliminate the risk of an immune system response, but magnets promised to be a more accurate and safer way to direct their creations around the body — with no need to convince a skeptical public that their tool isn’t a dangerous pathogen.
The Heart of the Problem
Heart disease is the world’s largest public health problem, responsible for killing more than 375,000 Americans each year — more than any other cause of death. Medical research has raced to keep up with treatments as the number of cardiovascular operations and procedures has grown by nearly a third to 7.6 million between 2000 and 2010, according to statistics compiled by the American Heart Association.
The most common type of heart disease, atherosclerosis of the coronary arteries, happens when the vessels that supply blood to the heart become narrowed by a buildup of cholesterol and plaque on their inner walls.
If those arteries become blocked suddenly, a heart attack results. But for about 10 percent of patients, blockage builds up slowly, and an individual may walk around for several months or more with a fully blocked artery, experiencing fatigue and chest pain as his heart slowly starves for oxygen.
This slow-motion heart attack is known as chronic total occlusion, and it’s the condition that most interests the researchers assembled by the Daegu Gyeongbuk Institute.
It used to be that the only treatment for a totally blocked artery was open-heart bypass surgery. In the past few years, cardiologists have had increasing success using angioplasty and stents to treat total occlusions. Angioplasty pushes out the blockage by expanding the artery with a balloon-like probe, while stents are mesh-like structures placed inside blood vessels to create a channel for blood to flow through.
But overall success rates for treating total blockages using stents and angioplasty can vary, depending on the condition of the patient and the skill of the surgeon, from 60 percent to 85 percent — far below the 98 percent success rate that can be achieved in patients with partial blockages. And blockages are known to recur inside stents 10 to 20 percent of the time, with many patients needing another procedure within a year of insertion.
That’s too many failed procedures, especially for an organ as important and delicate as the heart.
This is where micro and nanorobotics comes in.
The tools being developed by the Daegu Gyeongbuk team promise to be less invasive than a stent placement, and could also cut down on the risk of post-surgery complications. Many stents are made of metal that become part of the permanent wall of the blood vessel and sometimes trigger the development of scars or clots at the stent site. The beads that Kim has developed are made of materials that won’t trigger an immune response and that will degrade in the body without leaving a trace or damaging the integrity of the artery wall.
Mile Marker One
The crucial test for the Daegu Gyeongbuk team will be to successfully translate lab results to human studies. Kim’s microswimmers will be first tested in mice in 2016 and then in increasingly complex mammals over the next few years.
Kim has already shown that the technology works “in the glass” — that is, in a controlled liquid environment. In videos produced in his lab, microswimmers are filmed conducting small orchestrated tasks like moving toward targets and mimicking pre-set patterns and shapes, such as the outlines of the letters D and U (a microscale nod to Drexel University).
Inside the surging arteries of a human body, however, fluid dynamics are relatively chaotic.
“We have a lot of trial and error to do, but I’m excited,” says Kim.
It’s a goal with a huge payoff. Nanobots have potential beyond just treating blocked arteries. If they can be successfully steered inside the body, they could bring unprecedented precision to many medical treatments.
Kim is studying ways to make microswimmer beads move in swarms and work together toward a common goal, such as delivering doses of drugs in effective quantities to a target site in the body. For instance, after they’ve helped to clear an artery, his microswimmers could disperse and deliver anti-coagulant medication directly to the affected area to prevent clotting.
Another possibility is that medicated beads could be injected close to a disease site in the body and moved to the precise target.
“For cancers that have antigens, microrobots could deliver the antibodies right to the tumor site,” Kim says. “Or the structure and movement of the microrobots could allow them to penetrate through the cancer membrane, vastly increasing drug effectiveness.”
Kim is able to move a microswimmer with a margin of error of less than 2 microns — which would be valuable in surgeries where precision and nimbleness are essential, such as brain operations.
“Given what we’ve shown so far, nanorobots could be deployed and not damage a single neuron,” Kim says.
Kim hasn’t stopped seeing his dream up on screen in the years since Dennis Quaid landed a tiny craft on Martin Short’s optic nerve in “Innerspace.” Shows like “Arrow,” “Futurama” and movies like “GI Joe” have taken the mental leap that Kim hopes to make a reality.
“‘Fantastic Voyage’ came out 50 years ago,” Kim says, referring to the 1966 movie about a submarine shrunk small enough for its crew to repair brain damage — a poster of which he keeps on his computer for inspiration. “And to realize this science fiction dream in real life is what we’re working for.”