
_Liang Oscar Qiang
Qiang is a research assistant professor. Peter Baas is a professor, both in the Department of Neurobiology and Anatomy at Drexel’s College of Medicine.

_Peter Baas
Baas is a professor in the College of Medicine and director of the graduate program in neuroscience.
Scientific dogma holds that the protein tau stabilizes microtubules within brain cells — but new research suggests just the opposite. Tau’s actual role in the neuron is to allow microtubules to grow and remain dynamic, according to Liang Oscar Qiang, the study’s lead author and a research assistant professor in the College of Medicine.
This is critical because both the stable and dynamic regions of the microtubule must be present in the brain for successful cognitive function.
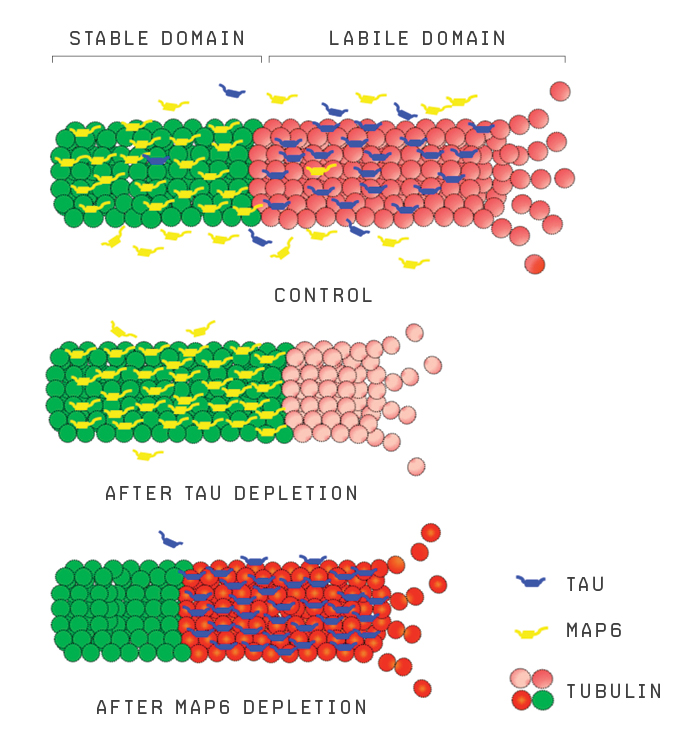
TAU-ISM
Tau and MAP6 help regulate the stable (green) and dynamic (red) parts of a neuron’s microtubules. Depleting tau from cultured rat neurons led to greater stability of the neuron’s microtubules.
Tau is one of the hallmark proteins of Alzheimer’s disease. In the diseased brain, tau breaks away from microtubules and forms neurofibrillary tangles, blocking nutrient transport inside of neurons and eventually killing them.
Drugs that affect microtubule stability are currently under investigation as potential therapies for Alzheimer’s, because it is nearly universally accepted by the scientific community that the role of tau is to stabilize microtubules in neurons of the brain, specifically in nerve fibers called axons.
The implication of this new discovery is that microtubule-stabilizing drugs currently in clinical trials may not be effective in treating Alzheimer’s and other tau-based neurodegenerative diseases, says Peter Baas, a professor in the Department of Neurobiology and Anatomy at the College of Medicine and the study’s principal investigator.
