
_Christos Katsetos
Katsetos is a professor of pathology and a research professor of pediatrics and neurology in the College of Medicine and a neuropathologist in the Section of Neurology and the Pediatric Neuro-oncology Program at St. Christopher’s Hospital for Children.
Being diagnosed with a diffuse glioma is akin to getting a death sentence. Current treatment options for this type of brain cancer are ineffective.
“These tumors cannot be adequately resected by surgery and do not respond to chemotherapy or radiotherapy,” says Christos D. Katsetos.
Cancer is the leading cause of non-accidental death in children, and brain tumors make up approximately 20 percent of childhood cancers. Gliomas account for the majority of primary brain tumors in children and adults, and even low-grade diffuse gliomas are potentially malignant and invariably lethal.
“Despite many recent advances in the genomics of brain tumors, there is no effective treatment for diffuse gliomas in general and glioblastoma in particular, necessitating an urgent mandate for new innovative treatment strategies,” Katsetos says.
Katsetos’ research focuses on diffuse gliomas in children because, at this time, there is no cure. He has unraveled key cellular mechanisms explaining why these cancers grow and spread within the brain and has proposed a possible way to halt tumor growth and invasion.
According to Katsetos, in addition to being resistant to chemotherapy and radiation therapy, “diffuse gliomas can’t be surgically removed because of the manner by which they violate the brain.” These tumors spread in a highly infiltrative fashion and don’t have borders, so they can’t be removed completely through surgery.
Tackling the grim prognosis of diffuse gliomas and glioblastoma in children and adults has been a life-long crusade for Katsetos as part of a world-wide collaborative effort. Toward this goal, Katsetos has collaborated with Pavel Dráber and Eduarda Dráberová from the Institute of Molecular Genetics at the Academy of Sciences of the Czech Republic in Prague; Maria Kavallaris from the Children’s Cancer Institute Australia; and Peter W. Baas, professor of neurobiology and anatomy and director of the Graduate Neuroscience Program at Drexel’s College of Medicine.
LETHAL_THREAT
Cancer is the leading cause of non-accidental death in children. Brain tumors make up about 20 percent of childhood cancers.
Katsetos and coworkers hypothesized that if they could elucidate how these tumors grow and invade the brain, they could potentially abrogate tumor growth.
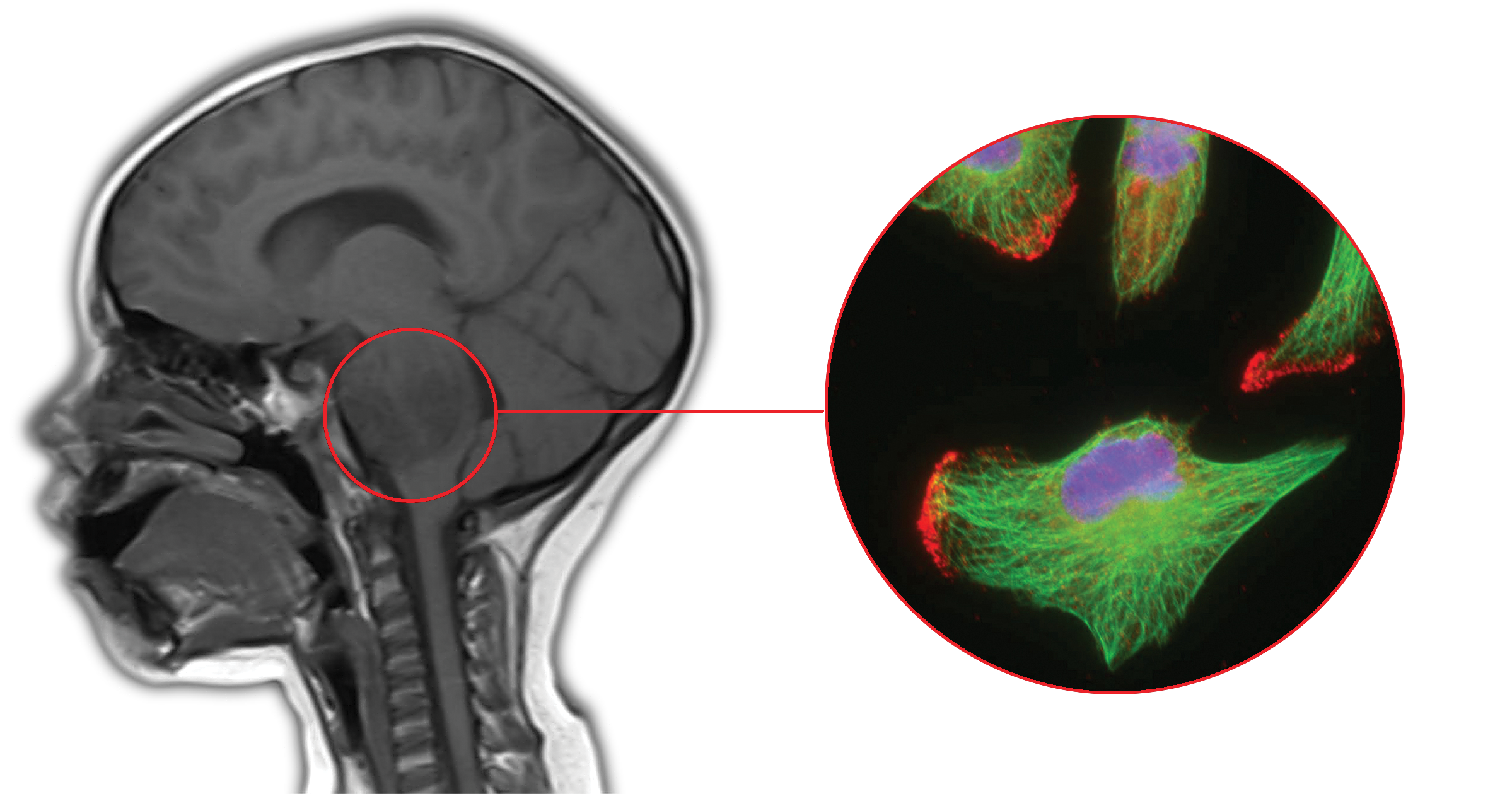
Katsetos’ research, which has been supported by grants from the St. Christopher’s Foundation for Children and the Philadelphia Health Education Corporation-St. Christopher’s Hospital for Children Reunified Endowment, led him to the study of subcellular structures of the cell’s backbone called microtubules.
In normal cells microtubules are an important part of cell life. They play both structural and functional roles in intracellular transport and trafficking, cell organization, cell division and cell movement. Without them, cells would not function.
But when microtubules in cancer cells are altered, they become unstable, which leads to wild growth and invasive spread of gliomas throughout the brain. One principal strategy used in the treatment of malignant tumors has been to disrupt the integrity of microtubules to prevent or curtail mitotic cell division.
Katsetos and his colleagues have previously shown that combined increased expression of regulators of microtubule formation and dynamics may define cancer cells with aggressive and invasive behavior. They are now attempting to disrupt the dynamically unstable microtubules of cancer cells, first in the lab and then in an animal model. Targeting key microtubule proteins is a novel treatment strategy aimed at slowing down tumor cell invasion given that microtubules are involved in tumor cell motility.
“It seems that silencing or knocking down genes encoding proteins associated with microtubules, such as the microtubule-severing enzyme spastin, mitigates or even halts tumor cell invasion, which is a promising step,” says Katsetos.