It was Aug. 15, 2020, five months into the lockdown, and Garth Ehrlich was desperate to get away.
“I hadn’t been out of my condo and couldn’t take it anymore,” recalls Ehrlich, who is a professor of microbiology and immunology, and otolaryngology, in Drexel’s College of Medicine. He loaded up his car and hit the road for his friends’ farm in the Midwest, where he planned to relax for his first vacation in six months.
“And then,” he recalls, “I got a call from the chair of my department.”
Chuck Cairns, the Walter H. and Leonore Annenberg Dean of the College of Medicine and senior vice president for medical affairs, wanted Ehrlich and his group, along with Brian Wigdahl, professor and chair of the Department of Microbiology & Immunology, to set up a SARS-CoV-2 testing lab, and he wanted it operational in six weeks. But there was no lab space, instruments, personnel, certified systems or accreditation to fulfill this request, which meant the clock was really ticking.
Still driving, Ehrlich began to make calls. He spoke to his lab manager and placed requests to find space on the University’s campus in Philadelphia. By the time he reached his friends’ farm, he was talking to manufacturers to get price quotes for lab instruments.
The lockdown retreat became a working vacation that changed the next few years of Ehrlich’s life. The facility he began assembling that day became the Pathology Diagnostics Laboratory (since renamed Drexel Medicine Diagnostics Laboratory), which would go on to process up to 10% of all polymerase chain reaction (PCR) tests in Philadelphia and 10% of all SARS-CoV-2 sequencing statewide. The lab made it possible for students to return to campus a few months later, helped Philadelphia and Drexel partners monitor new variants, and transformed several academic careers.
Two years later, with the pandemic in retreat, the lab’s creators have repurposed it as a new commercial testing service. This time, though, they have more than six weeks — and they have their own footsteps to follow.
STEP 1: Take Stock of What You Have
At the time Ehrlich got his call, Drexel was teaching remotely and the streets of Philadelphia had gone quiet. The University’s Student Health Center was offering limited COVID testing for certain groups (Dragons working in clinical settings, for example) that it paid a private company to analyze.
For everyone’s safety, Drexel leaders wanted students and employees to remain remote until the University had testing capability with same-day results of its own.
Ehrlich had some experience creating a clinical molecular diagnostics lab for the University of Pittsburgh’s pathology department, but that was about 25 years ago. “It wasn’t something that I was planning on ever doing again,” he admits, “but I was happy to do it because I did feel that we were doing something good for the University and the community.”
For the undertaking, he partnered with Azad Ahmed, a long-time colleague from his University of Pittsburgh days who helps him run the Genomics Core Facility (GCF), Drexel’s gene sequencing and bioinformatics service core. The GCF houses instrumentation platforms for DNA and RNA sequencing — so Drexel could analyze positive SARS-CoV-2 tests and identify the variant.
But Drexel would need a way to collect those samples, extract the RNA from them, convert the RNA to DNA for sequencing, monitor the sample at every step of its testing and surveillance journey, and ultimately notify individuals of a positive or negative result.
“We had no space, no equipment, no personnel, no protocols, no nothing,” says Ehrlich.
What they did have was Jaroslaw “Jarek” E. Król and Donald “DJ” Hall (PhD ’20).
Król was the Center for Genomics lab manager and the first person Ehrlich called. An assistant professor in the Department of Microbiology and Immunology, Król played a major role in setting up the lab and served as lab mentor to Hall, who had just graduated from Drexel with a chemistry doctoral degree in summer 2020. Hall had been planning to do a research postdoc with Ehrlich, but when the pandemic shut down the University, he took on the role of operations director of the new coronavirus lab.
“Originally, I’m a medicinal chemist, not a clinical chemist, so I had never worked in a clinical lab,” says Hall. “But I volunteered to help because this got me out of the house, which was something I really needed.”
In a matter of hours, they had assembled a lab startup team. That was the easy part. Tick, tick, tick…
Step 2: Source All of the Necessary Parts
Ehrlich’s unit had been one of the first to close at the University when the pandemic arrived. Now it was the first to reopen.
The team decided that they would take over Drexel’s former Clinical and Anatomic Pathology Labs, on the 5th floor of the New College Building in Center City, and combine that with Ahmed’s GCF labs on the 17th floor. The shuttered lab hadn’t been used in almost a year, a casualty of the 2019 bankruptcy and closure of Hahnemann University Hospital, which had been the lab’s main source of specimens.
Alone in the vacant downtown building, Ahmed, Hall and Król sat through demonstration after demonstration of devices from various manufacturers, while Ehrlich, who was at high risk, advised from home. The team selected supplies and equipment from two different companies, to offset any possible supply chain lags.
TESTING_TIMELINE
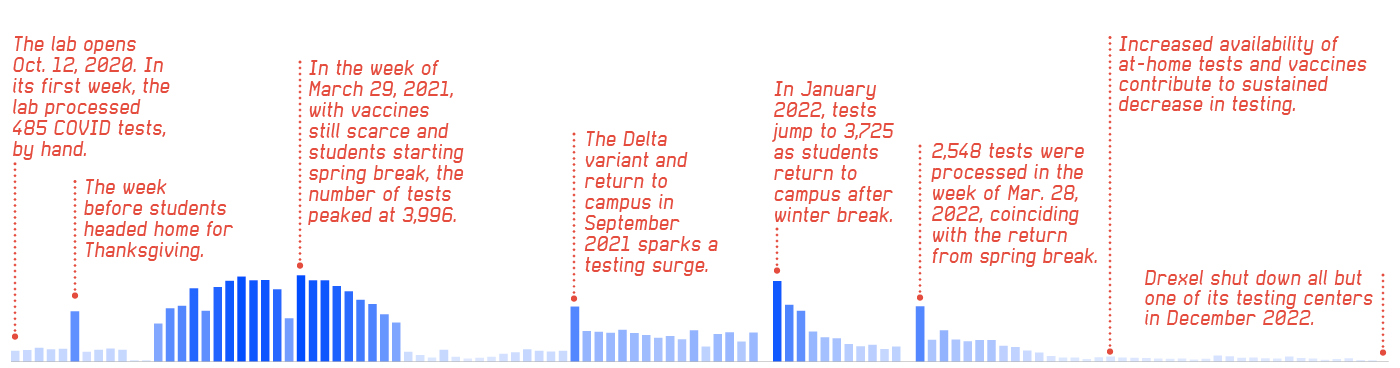
The lab’s workload waxed and waned with the cycles of Drexel’s quarter system.
One of the companies, Tetracore, already had an immunology research collaboration with the College of Medicine and had been recommended to the College of Medicine’s Dean Cairns by MyOwnMed, a digital platform that he worked with to create Drexel’s symptom-tracking app, the Drexel Health Checker, in April 2020.
“We were able to leverage ongoing research partnerships to immediate impact through these incredibly important connections with MyOwnMed on the software development side and Tetracore on the testing supply side,” says Cairns. “It resulted in a pretty remarkable collaboration.”
Drexel spent about $2 million of the federal coronavirus relief funding it received to equip the lab. A few weeks into the six-week deadline, things were getting checked off a massively important to-do list.
But there was still a lot to do and even more to learn.
One very large hurdle they faced was accreditation. Most of the team were scientists whose work had never required them to link their samples to patient information. But now they would need to handle “real people” specimens and issue test results.
“To do that, there are two different types of accreditations you need to run the lab: You need a clinical lab permit from the state, and you need a pathologist in the lab as a medical director,” says Professor Cheryl Hanau, who is the Richard Shuman, MD, Chair of the Department of Pathology & Laboratory Medicine. “Fortunately, there was one pathologist left at Drexel: me.”
Hanau had been the medical director of the former Anatomic Pathology Lab when Hahnemann closed and everything became “extinct,” as she remembers it. After the hospital’s bankruptcy, the lab closed, but Hanau decided to stay at Drexel on a reduced salary as a professor and academic chair of the Pathology Department, with no clinical responsibilities and no reason to work as part of a lab … until now.
Thankfully, her name was still on the lab’s two accreditations and state permit. Thankfully, she had maintained an active Pennsylvania medical license. Thankfully, she had stored all the lab’s processes and documentation. Thankfully, she was just as willing as everyone else to jump in and help.
Step 3: Put the Pieces Together
Through lots (and lots) of phone calls, Hanau learned that Drexel could reactivate its license from the Clinical Laboratory Improvement Amendments (CLIA) and its Clinical Lab Permit from the Commonwealth of Pennsylvania. Drexel would also need a new accreditation certificate from the College of American Pathologists (CAP), which is authorized by CLIA to ensure the quality of the lab through inspections and proficiency testing.
“When I called CAP, the first thing they asked was what state the lab was in, because each state has different rules for accreditation,” remembers Hanau. “When I said ‘Pennsylvania,’ the person on the phone said, ‘Oh, I’m so sorry.’”
She learned that Drexel’s lab permit was valid and could be reactivated, but that the state would have to inspect the space before it could function — and that could take up to a year due to an inspector shortage.
In the meantime, the team had plenty left to do. The lab needed a new laboratory information system to track specimens, because it had previously relied on Hahnemann’s system. Joshua Earl, an assistant professor in the Department of Microbiology & Immunology in the College of Medicine and GCF’s head of bioinformatics, stepped in to create a software system to support the lab’s operations.
The lab also needed an electronic health record — also formerly supplied by Hahnemann for hospital-based specimens — to order and schedule tests as well as to report the results to the patient and other stakeholders, such as the city and Drexel’s contact-tracing team. They modified the Drexel Health Checker app that Cairns had created with MyOwnMed; now, the app could be used to schedule a test and receive results. Behind the scenes, the app would generate a label for the sample to be tracked during the testing process.
The necessary regulations, standard operating procedures, worksheets, reporting forms and other rules had all been saved by Hanau, but everything needed to be updated for the new lab.
“I’ve never heard of this happening anywhere else before,” Hall says of the process. “We went from nothing to running samples for surveillance in just a few weeks, to reinstating our CLIA license and running diagnostics and becoming CAP accredited in just over a year.”
CAP accreditation aside, the lab was ready to begin testing on Oct. 1 (and to begin surveillance on Oct. 11), on schedule. It opened as the Pathology Diagnostics Laboratory with lab personnel running tests by hand at first, uncapping and transferring samples themselves, until automated machinery arrived later.
“Three months after we ran our first test on Oct. 1, we went from doing a handful to over 1,000 a day and over 5,000 a week,” says Ehrlich.
The CLIA and the state licenses were approved in December (CAP accreditation came later). By then, Drexel had opened three COVID testing sites on campus, staffed with students and faculty from the College of Nursing and Health Professions and the College of Medicine.
With that, the University could safely invite Dragons back to campus in January 2021.
Step 4: Ramp Up and Step Up
While Omicron raged in the winter of 2021, the Pathology Diagnostics Laboratory was running up to 1,200 tests a day.
With variant fears running high, others in the city took notice of Drexel’s lab.
Joshua Chang Mell, an assistant professor in the College of Medicine, and longtime collaborator Paul J. Planet, an assistant professor at the University of Pennsylvania and co-director of the Children’s Hospital of Philadelphia (CHOP)’s Microbial Archive and Cryocollection, had wanted to use their experience sequencing bacterial pathogens to help in some way.
Near the end of 2020, they had submitted a grant to the Centers for Disease Control and Prevention (CDC) to track variants by sequencing samples collected at CHOP. That collaboration was funded and expanded to include the Philadelphia Department of Public Health (PDPH), when they began sequencing specimens collected at some of the city’s sites, like public schools, nursing homes and prisons.
“We went from nothing to running samples for surveillance in just a few weeks, to reinstating our license and running diagnostics and becoming accredited in just over a year.”
Donald “DJ” Hall (PhD ’20)
Lab director
But while the PDPH could run diagnostics tests, it didn’t have a sequencing lab. And while CHOP had sequencing capabilities, Drexel’s lab was superior at extracting RNA, Planet says, so Drexel eventually took over the extraction and sequencing of samples.
“Before the pandemic, I had worked with all of the Drexel people as collaborators and researchers, and I saw that they did a really excellent job at setting up that lab, so it didn’t really make sense that we weren’t working together when we were doing these parallel things,” says Mell, who jokes that he and Planet were the lab’s first customers.
“We found that the infrastructure for extraction led to better results if we brought it to Drexel,” says Planet. “It certainly takes more time to transfer samples between the two campuses, but it actually winds up being better overall quality and is more efficient.”
The CHOP-collected specimens would go to Drexel for extraction, back to CHOP to be prepared in libraries, back to Drexel for sequencing, and then back to CHOP for final analysis. The entire process took a few days to a week.
The “coolest period of time in the collaboration,” Mell recalls, was the race to find Omicron in the city. The new variant was finally collected at CHOP’s drive-through site in early December 2021 and sequenced by Drexel a few days later. In mid-December, Omicron was detected in less than 1% of their samples, but within a few weeks it made up 90% of positive COVID tests.
“We had this really crazy period of time where everything was moving at factory speed,” says Mell.
Demand for testing was so high then — around Drexel’s winter break, too — that the lab went from reporting diagnostic results within eight hours to 24 hours.
But this wasn’t the Pathology Diagnostics Laboratory’s first time weathering a surge, and as cases rose, the staff developed a time-saving trick. They discovered they could use their stockpile of older PCR tests, which had been made obsolete by the Delta variant, to pre-screen samples for a gene known to be part of the Omicron spike protein, which allowed them to quickly identify the new variant without having to run DNA sequencing.
Step 5: Survey What You Have Created
From fall 2020 to fall 2022, the Pathology Diagnostics Laboratory ran about 130,000 diagnostic tests, as well as thousands of sequencing procedures for the University, CHOP and the city. At one point, the lab produced 5–10% of the sequencing for all of Pennsylvania. The lab also saved Drexel considerable money: its cost to run a test was $21 compared to $120 charged by vendors.
The team’s experience, connections, resourcefulness — and willingness to drop everything to take on the task — was an enormous service during the pandemic. The lab enabled a safe return to campus, allowed student-athletes to participate in group sports, provided gold standard testing when tests were scarce, and informed the University’s safety decisions. During the Omicron surge, data from the lab convinced Drexel to modify its winter academic schedule, for example.
“We can provide a service that’s state of the art, agile and adaptive. That’s the experience and expertise our people learned from COVID.”
Chuck Cairns
Dean, Drexel College of Medicine
“For the first year and a half of the lab’s existence, we were constantly racing just to meet the marks,” says Ehrlich. “It was really just a heroic effort by the team to meet these ever-increasing targets with both sophistication and numbers.”
The lab’s work began to wind down as at-home tests became more available and policies for mandatory gateway testing changed. By summer 2022, the lab was testing only 70 to 80 samples a month. In December 2022, Drexel closed all but one of its testing centers.
Step 6: Keep Adapting
In July 2022, Philadelphia opened its own sequencing and bioinformatic lab with a CDC grant, making it one of only four public health labs of its kind in the country.
When the city was assembling its lab, its employees contacted the Pathology Diagnostics Laboratory and the GCF for tours, demonstrations and consultations.
The city ended up buying the same system and platform for reading sequences, the same automated pipettors to put samples into tubes and the same liquid handlers for transferring the samples. One vendor even called Hall to thank him for the referrals.
“Drexel is really on the forefront of a lot of stuff that we can learn from,” says Bernadette Matthis, director of the city’s new Public Health Laboratory. “We had a good foundation for what direction to proceed in.”
As for the Pathology Diagnostics Laboratory, it has outlasted the pandemic.
With a luxurious 12 months to prepare, the team is transforming it into a full-service diagnostic lab to test for infectious diseases, genomics and toxicology — allowing it to serve as both a research resource and a revenue source for the University.
Its new name is the Drexel Medicine Diagnostics Laboratory, and it is slated to open in summer 2023.
“Nothing ever stays the same and the paperwork is always constant, but there are lots of opportunities to learn,” Ehrlich reflects. “The lab’s expansion will leverage the University’s investment in time, personnel and instrumentation for building up the lab.”
Cairns, the College of Medicine dean, hopes the new lab can serve Drexel’s colleges and schools and offer a chance for Drexel to branch out.
“If you think about research with impact as innovation, it shows that we have the innovation ability to do it,” he says. “We proved we could do it in COVID with an unknown agent. Imagine if we do it with a known genetic target. We can provide a service that’s state-of-the-art, agile and adaptive. That’s the experience and expertise our people learned from COVID.”