
_Peter Baas
Baas is a professor in the College of Medicine and director of the graduate program in neuroscience.
Army veteran Lynn Santosuosso donated her blood to science last spring. From a Boston University lab, her cells were sent to the school’s Center for Regenerative Medicine, where scientists inserted into them four carefully chosen genes.
The modification transformed the cells, reversing them back to their infancy and making them ripe with research potential.
Laboratory-grown stem cells — called induced pluripotent or iPS cells — are adult cells that have been genetically reprogrammed to behave like embryonic stem cells. They not only proliferate indefinitely inside a petri dish, but can also differentiate into any type of cell in the human body.
“They all were coming back with these unexplained symptoms. Fewer than 10 percent of the veterans we saw were diagnosed with post-traumatic stress disorder, so clearly something else was happening.”
From Boston, Santosuosso’s iPS cells then journeyed 300 miles to Drexel’s College of Medicine in Philadelphia. There, a team of neuroscientists altered them once again — this time, into neurons.
The Drexel researchers believe these modified cells could hold answers to the debilitating cluster of symptoms that have plagued Santosuosso and her peers since they returned from the first Gulf War in 1991. It is estimated that 250,000 of the 700,000 veterans deployed in the conflict suffer from what is now known as Gulf War Illness.
“We see this as an urgent situation; these veterans have been suffering for more than 30 years,” says Peter Baas, a professor in the Department of Neurobiology and Anatomy, who is leading the Gulf War Illness research efforts at Drexel. “Until now, a real challenge for the research community was developing models to study this disease.”
The pluripotent stem cell lines offer scientists, for the first time, the opportunity to identify the neurological underpinnings of Gulf War Illness and to develop treatments to reverse it.
unhealthy winds of war
After 10 years in the Army Reserve, Santosuosso was called up for active duty in November 1990 during Operation Desert Storm. She was overseas for six months, stationed north of King Khalid Military City in Saudi Arabia.
Outside her tent, the sound of air raid sirens and chemical alarms frequently filled the air. But long-term environmental health risks were the last thing on her mind.
“The only thing you’re thinking about is what your mission is and doing your job,” the 56-year-old engineering technician says now.
When she returned home to New England, Santosuosso began forgetting where she put things and losing her words mid-conversation. She experienced migraines, an unrelenting cough and difficulty sleeping. Often, she woke up in the middle of the night unable to breathe.
Around the same time, Kimberly Sullivan, who was training as a neuropsychologist at the VA Boston Healthcare System, was noticing similar cognitive problems in the veterans whom she evaluated.
“They all were coming back with these unexplained symptoms,” says Sullivan, now a research assistant professor at Boston University.
Veterans complained of fatigue, headaches, joint pain, indigestion, insomnia, dizziness, respiratory disorders and memory problems. “Fewer than 10 percent of the veterans we saw were diagnosed with post-traumatic stress disorder, so clearly something else was happening,” she says.
Officials in the Department of Defense (DoD) questioned whether the strange symptoms could be tied to chemical pollutants that had affected the veterans’ central nervous system. The DoD subsequently funded a study for Sullivan and a research team to conduct follow-up interviews and cognitive evaluations with any men and women who had handled pesticides during the war.
A series of studies and investigations later revealed that the Gulf War veterans were exposed to combinations of toxic pesticides — used, among other purposes, to treat uniforms, and as insect repellent on the skin — as well as pyridostigmine bromide (PB) pills and anthrax vaccines. The military had prescribed the “PB” pills using a waiver from the Food and Drug Administration, which allowed the troops to take the otherwise non-approved prophylactic as prevention against nerve agent poisoning. In 1996, reports uncovered that the United States’ 1991 bombings of an ammunitions storage depot in Khamisiyah, Iraq, had unintentionally released a wave of sarin nerve gas. The gas had drifted southward more than 300 miles to Saudi Arabia — where 100,000 American troops were stationed.
“They were exposed to a toxic soup of chemicals,” Sullivan says. “We didn’t understand then what a mixture like that could do. The thought was that if there wasn’t a major poisoning event, then there wouldn’t be chronic health outcomes. We now know that is not the case.”
Since those early revelations, Sullivan has dedicated her research to finding out more about the origins of Gulf War Illness and how to help veterans affected by it.
Santosuosso connected with Sullivan’s research group in the late ’90s and has been participating in studies at Boston University ever since. But despite a willingness to volunteer her body for science, she has received little effective treatment for her symptoms, which she believes have grown worse over the years. In three years, she’ll be able to retire from her job at the Portsmouth Naval Shipyard in Maine.
“Hopefully I can continue working that long,” she says. Between fibromyalgia, chronic fatigue and memory issues, she says it’s becoming more difficult to remain productive. “It is frustrating as hell some days.”
A scientific metamorphosis
Drexel’s Peter Baas knew little about Gulf War Illness 10 years ago.
He came to study the disease through his career investigating microtubules — hollow cylinders that provide important structure and shape to a cell, and also act as railways that transport organelles throughout cellular cytoplasm. When neurons have microtubule abnormalities, a lot can go wrong in the brain and central nervous system. In Alzheimer’s disease, for instance, chemical changes cause a protein called tau to detach from microtubules, making the neurons dysfunctional.
In 2006, the DoD was offering grants for research scientists to study Gulf War Illness. Baas applied for and earned the funding, hypothesizing that microtubule-related proteins could play a role in the neurodegenerative disease. He was soon invited to take part in the Gulf War Illness Consortium, funded by the DoD and led by Sullivan. The consortium, which is still active today, consists of researchers from 10 institutions working to solve the mystery of Gulf War Illness through clinical and animal studies.
But although living patients and animals provide a range of opportunities to study the disease, the researchers’ lack of postmortem brain tissue created barriers for testing mechanistic hypotheses and screening therapeutic compounds.
“I think what became more and more obvious, is that the DoD was getting a little dubious about the use of rats and mice to study Gulf War Illness, because there seemed to be some genetic and epigenetic factors to the disease,” Baas explains. “Why did some soldiers get sick, while others who were exposed equally, did not? Was there some kind of predisposition to the disease?”
Not only is it difficult to study genetic factors of disease within animals, but rats are also poor models for studying neurodegeneration more generally.
“Human neurons are just not the same as the cells in rats or mice,” Baas says. “For example, the tau protein that goes awry in Alzheimer’s makes neurofibrillary tangles. In rodents, the tau doesn’t make those same tangles. So, certain key disease mechanisms are not re-capitulated in animals very well.”
A breakthrough in the study of human cells occurred in 2006, thanks to Nobel Prize-winning research by Japanese scientist Shinya Yamanaka.
It was Yamanaka who first “reprogrammed” adult stem cells into induced pluripotent stem cell lines. Though originally intended for clinical application, these cells have become a critical tool for biological research.
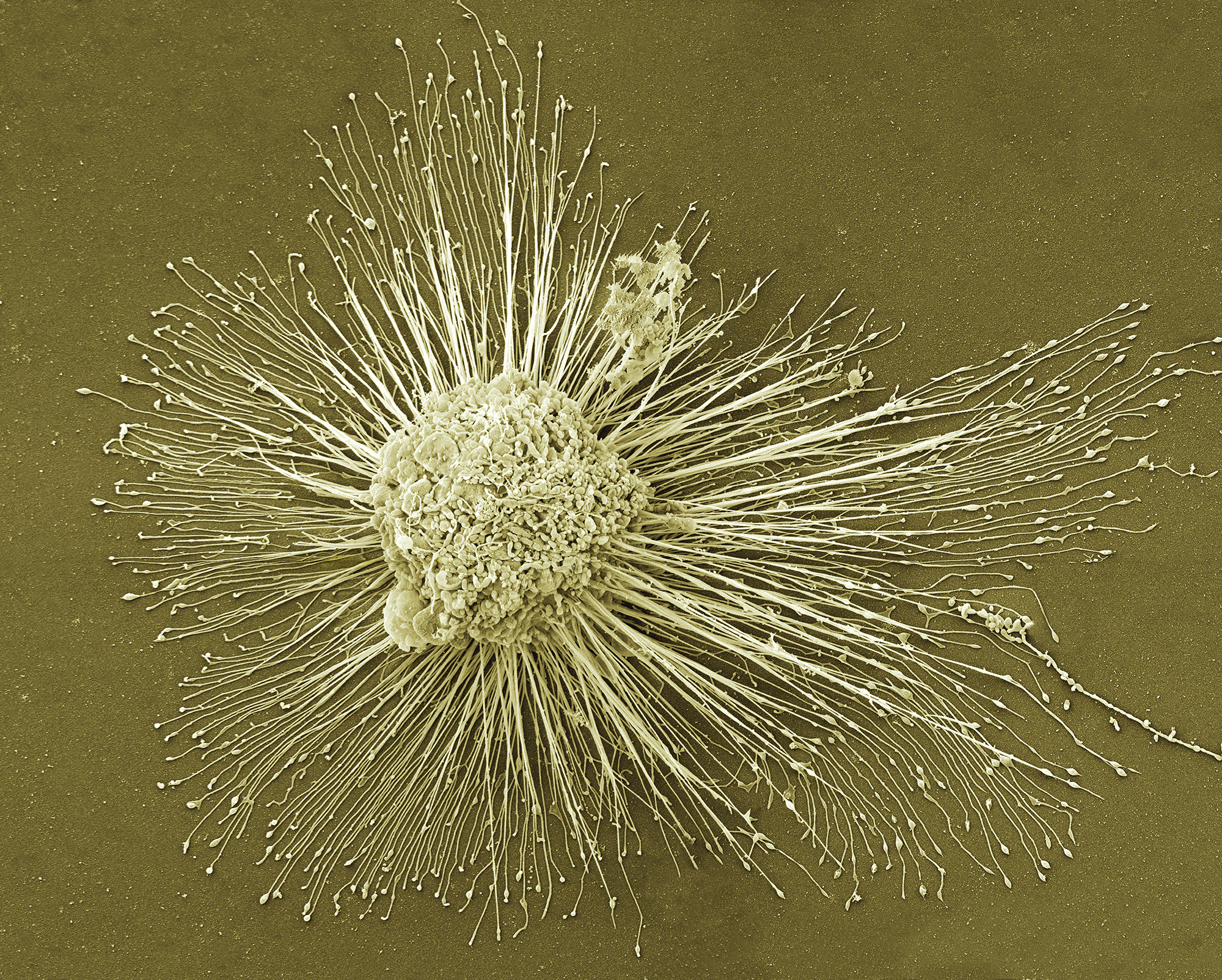
IPS_Cells
Laboratory-grown stem cells — called induced pluripotent or iPS cells — are adult cells that have been genetically reprogrammed to behave like embryonic stem cells. Drexel researchers believe these modified cells could hold answers to the debilitating cluster of symptoms that have plagued many veterans since they returned from the first Gulf War in 1991.
Now, the Gulf War Illness researchers will be the first team in the world to study blood cells obtained from 300 veteran volunteers and modified using Yamanaka’s technique. With the correct combination of growth factors, scientists can manipulate the induced pluripotent stem cells into any type of cell they wish to investigate. And, because pluripotent cell lines are essentially immortal, they can be harvested for an unlimited number of studies. The reprogrammed stem cells will be housed in a biorepository at Boston University and made accessible to any researcher in the world.
Most importantly, the cells are derived directly from the patients affected by Gulf War Illness, which means they harbor the wide array of genetic factors that may contribute to the disease, says Liang “Oscar” Qiang, a research assistant professor in the College of Medicine.
“The big advantage of using patient-derived calls is that many diseases cannot be traced to just one gene mutation, but rather are due to the complex interactions of genes,” says Qiang, Drexel’s iPS cell expert. “Susceptibility of these diseases may also be based on epigenetic factors, which cannot be looked at in animal models. These cell lines will preserve the complete genetic composition of those affected by the disease.”
Treating a toxic world
In preliminary studies that used iPS cells (though not ones derived from Gulf War veterans), Baas and his research team have already identified a potential treatment for Gulf War Illness symptoms.
In one investigation, Drexel researchers asked whether elevated stress hormone levels could contribute to the effects of organophosphate (OP) chemicals — like the sarin used in pesticides and nerve agents during the war. To test their theory, the scientists treated cultures of neurons with an analog of sarin called diisopropyl fluorophosphate, along with cortisol. Within the neurons, the team was looking for deficits in the activity of microtubules.
Their results, published in the scientific journal Traffic in 2017, showed that even low levels of exposure to organophosphates can lead to lasting functional deficits. The sarin analog reduced the transport of mitochondria in the neurons, as well as the release of the neurotransmitter dopamine. Pre-existing levels of the stress hormone cortisol worsened these deficits even further.
The researchers were able to correct the deficits using Tubacin, a drug that preserves normal cellular function by restoring microtubule acetylation and correcting changes in microtubule structure. This suggests that microtubules could serve as a desirable structural target for Gulf War Illness treatment.
Surprisingly, the researchers also found that once they corrected the microtubules deficit, defects in dopamine release also markedly improved. Fluctuations in dopamine are thought to be connected to many of the neurological symptoms that Gulf War Illness sufferers face, including insomnia, cognitive problems and headaches. The study suggests that dopamine alterations after toxin exposure are in part due to changes in microtubules, and restoring microtubule function to a more normal state could help to alleviate symptoms.
“The fact that a microtubule-based therapy would correct the problem with dopamine release is very encouraging,” Baas says.
In future studies, the cells derived from Gulf War veterans will offer the researchers a clearer picture of how microtubule-based therapies might affect the illness. In addition to microtubules, they are investigating how other targets, such as tau proteins, may play a role in the disease.
The good news, Baas says, is that once a target is identified, it is likely that an FDA-approved drug already exists to treat the problem. However, the treatment would still likely have to go through vigorous clinical trials in order for the Department of Veterans Affairs to approve it for use on Gulf War Illness patients. Sullivan, Baas and the rest of the Gulf War Illness Consortium team are poised to begin these clinical trials once the most promising treatments are identified.
For Baas, finding a therapy that will help veterans like Lynn Santosuosso is the ultimate goal, but the possibilities for research don’t end there. The wide use of organophosphates as pesticides and herbicides across the world adds another layer of urgency to understanding the risks of ongoing chemical exposure. Extremely toxic chemicals are still used regularly in places like India and China, and organophosphates have also been used in public health applications in some countries, for example to combat West Nile virus. Not to mention, bioterrorism remains an ever-present threat.
“We’re living in an increasingly toxic world,” Baas says. “It’s likely that this kind of disease is going to repeat itself if we don’t educate ourselves to its causes, as well as how to prevent and treat it.”
