When it comes to Alzheimer’s disease, there is no denying the staggering numbers: It affects 5.4 million Americans and claims the No. 6 spot among the leading causes of death in the United States. It is the only one of the nation’s Top 10 killers that cannot be prevented, cured or slowed.
For those lucky enough to reach the age of 85, roughly half will develop the disease and likely die from its complications.
“If you live long enough, this is almost an inevitability,” says Aleister Saunders, Drexel’s senior vice provost for research, who has been studying Alzheimer’s disease since the ’90s.
Serious dollars have been spent in the hunt to better understand the disease — in 2015, Alzheimer’s and other dementias cost the nation $236 billion. By 2050, these costs could rise to $1.1 trillion, according to the Alzheimer’s Association. And these figures do not include the loss of wages for family caregivers, which typically equal or even exceed the cost of the health care.
Still, the disease’s etiology remains unknown. And the research dollars, which are only a fraction of that spent on diseases like HIV and cancer, are not enough, not by far.
Most research grants go toward studies based on the widely accepted hypothesis that the disease is caused by either intracellular neurofibrillary tangles or extracellular buildups of sticky beta-amyloid plaques in the brain, both of which are thought to interfere with communication between neurons and lead to inflammation, memory loss, cognitive decline and eventually death. It remains unknown, however, whether these processes cause the disease, or are merely symptoms.
Garth Ehrlich, an internationally recognized professor of microbiology and genomics in Drexel’s College of Medicine, is one of an increasingly vocal number of scientists who think Alzheimer’s research is headed in the wrong direction.
“Despite the billions that have been spent, we have no effective drugs, we have done nothing to change the natural history of the disease,” says Ehrlich. “This stands in stark contrast with the other chronic conditions that we deal with like cancer, stroke and heart disease. For Alzheimer’s we have nothing. The approach people are taking is not right.”
Some scientists now believe that chronic microbial infection of the brain is the culprit in Alzheimer’s disease.
Not-so-simple-cells
Bacteria may be single-celled organisms, but they shouldn’t be underestimated. In addition to forming biofilms for self protection and reproduction, they’re capable of combining into multiple large-ordered structures, analogous to the tissues of highly differentiated animals such as ourselves.
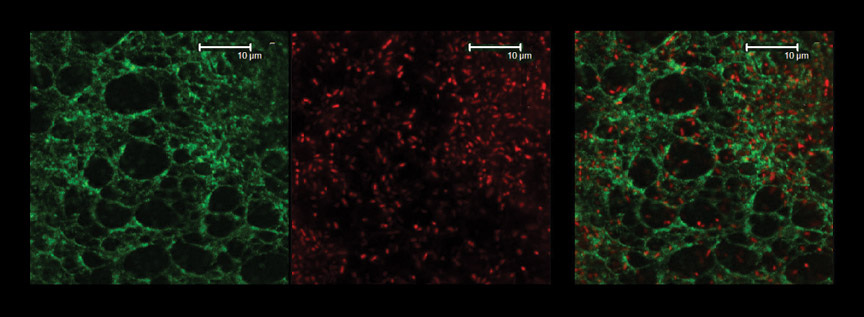
Highly organized extracellular structures built by the common opportunistic pathogen bacterium, Pseudomonas aeruginosa.
left regular hexagonal extracellular matrix that the bacteria form.
center actual bacteria (each point of red is a bacterium).
right superposition of the top two images which shows the bacteria embedded in the organized matrix that they have produced.
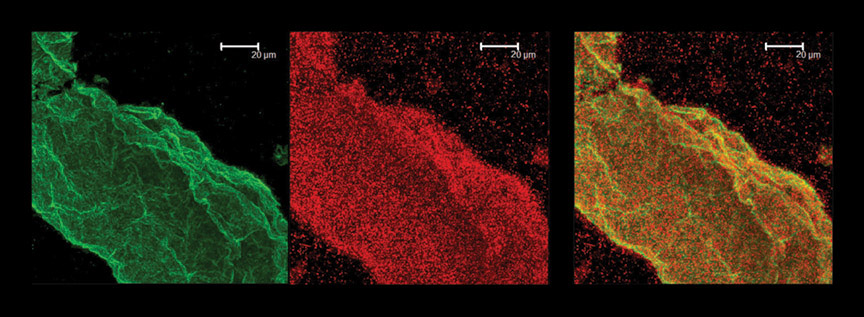
bacterially produced filaments
left matrix only
center bacteria
right bacteria embedded in the matrix they have extruded.
Chronic infectious disease happens to be Ehrlich’s wheelhouse. He and his team have spent years perfecting tools for studying how bacteria form multicellular and polymicrobial communities that can persist for years as deeply entrenched infections protected by extracellular matrices in the body.
Now they’re ready to test the Alzheimer’s chronic-infection hypothesis.
With brain samples supplied by the University of Arkansas, Ehrlich has designed a blinded study of post-mortem samples that uses a commercial pan-domain diagnostics assay and a second, more powerful, pan-domain assay newly developed in his lab, both of which can identify essentially all species of bacteria present within human tissue.
If the team confirms bacterial infections in the brains of the patients who died of Alzheimer’s, the findings could shift the research paradigm toward the development of tests for bacterial detection in the central nervous system and new treatments for combating chronic infections and inflammation.
It’s a theory that “deserves to be rigorously tested,” Ehrlich says.
All that’s needed is funding.
Biofilm_on_the_Brain
Drexel dermatologist Herbert Allen has experience studying how biofilms interact with immune cells in the skin and shares Ehrlich’s interest in the biofilm-Alzheimer’s hypothesis.
Allen, who is chairman of Drexel’s Department of Dermatology in the College of Medicine, showed that eczema may be caused by biofilms in a 2014 study. The study proposed that staphylococcal bacteria that live on the skin produce a biofilm that blocks sweat ducts, activating the immune system, which causes itching and irritation.
More recently, Allen published a small study in Neuroinfectious Diseases that investigates whether spirochetes in the brain could be operating in the same way to cause Alzheimer’s.
Allen and a team of collaborators examined seven post-mortem brains of Alzheimer’s disease patients, comparing them to 10 control samples from people who had died from other causes.
In each of the Alzheimer’s brains, the researchers saw what they believe to be biofilms. The researchers also identified a protein called TLR 2, which plays a key role in activating the body’s immune system.
Allen is a member of the camp that hypothesizes that the spirochetes enter the brain during a dental procedure or after a person contracts Lyme disease, and then spin out a protective biofilm. The body’s first responders try to clear the infection, but immune cells cannot penetrate the biofilm matrix that encases the organisms. Instead, the immune system destroys the surrounding tissue.
“So it’s neither the infection, nor the biofilm, nor the beta-amyloid that causes the disease, but the body’s own immune system,” Allen argues.
— Lauren Ingeno
‘A Moving Target’
Ehrlich has been studying the infectious causes of disease for three decades. He’s an expert on bacteria and how they communicate with one another, how they grow and spread, how they function; he’s spent his career marveling at how they’ve evolved over the last four billion years, including their adaptation to survive modern medical attempts to fight them.
One of the ways he believes bacteria cause the long-term ravages of Alzheimer’s is through their ability to form biofilms, which are slimy matrixes of living and dead bacteria cells bonded together by DNA, proteins and complex sugars that provide adherence to each other and to surfaces in the body. Biofilms congregate on mucosal surfaces; within spaces in the body like the sinuses and the cavities of the middle ear; and around foreign objects such as artificial joints and pacemakers. Think of the way plaque forms on teeth and harbors cavity-causing bacteria — the plaque is a biofilm.
Biofilm formation starts when a single cell attaches to a surface. It waits. Finds friends that have attached nearby. They link up, they form colonies, then form slime. Within the layers of slime, the innermost cells are able to ramp down into a metabolically dormant state that shields them from antibiotics.
They’re also able to swap genes amongst themselves to create new strains, “like sex for bacteria,” Ehrlich explains, only half jokingly. It’s a virulence phenomenon Ehrlich himself first postulated, known as the distributed genome hypothesis.
In one study of long-term infection conducted by Ehrlich in the early 2000s, his colleagues collected throat swabs from a child with chronic ear infections, so-named “Patient 19,” for seven months — and he found that one strain appeared over and over, but kept changing. “This one strain underwent 24 transformation events, replacing 8 percent of its genome with pieces of its buddies,” Ehrlich notes.
The ability to go dormant and to share genes, some of them progressively more resistant to antibiotics, makes bacteria living in biofilms very, very difficult to identify through cell cultures or to eradicate.
“Because the cells in a biofilm are transforming and exchanging genes and creating new strains, the body’s immune system is always chasing a moving target,” Ehrlich explains. “How do you combat something that is always changing?”
Ehrlich began studying the way bacteria communicate, collaborate and colonize into biofilms in 1996, when he befriended microbial ecologist Bill Costerton, known in the field as “the father of biofilms.” At the time, Costerton was director of the NSF-funded Center for Biofilm Engineering at Montana State University, which soon became the leading biofilm research center in North America. Late one night, Ehrlich was reading an article in Science magazine authored by Costerton that included the sentence, “Bacteria in biofilms can be very difficult to culture and are resistant to up to one thousand times the concentrations of antibiotics that can kill their free-swimming counterparts.”
A light blub went off, Ehrlich says.
He immediately recognized that Costerton’s biofilm paradigm would explain the contradictory results he had been seeing in chronic middle-ear infections in children. He and his colleague, an ear-nose-and-throat surgeon named Chris Post affiliated at the time with the University of Pittsburgh and the Children’s Hospital of Pittsburgh, had found evidence of live, metabolically active bacteria in the middle ear canals of children with chronic effusions. Puzzlingly, the bacteria could not be cultured, yet survived repeated courses of antibiotics.
Ehrlich contacted Costerton, and together they forged a fruitful collaboration resulting in a series of papers demonstrating that all of the common bacteria associated with sore throats and other childhood maladies could form biofilms in the middle ears of both chinchillas and children. Through subsequent research, they were eventually able to generalize these findings to all chronic mucosal infections and eventually to all chronic bacterial infections.
Ehrlich went on to devote years to studying biofilm science and perfecting DNA tests to prove the presence of hard-to-culture bacteria. He has published more than 100 papers and two textbooks proving that many chronic inflammatory conditions are actually biofilm infections.
About six years ago, he began to consider the possibility that the inflammation associated with Alzheimer’s might also be a symptom of an established infection that had traveled from the sinuses to the brain.
Biofilms that form in middle-ear mucosa and on implanted devices like pacemakers and artificial joints can be removed surgically, but what about biofilms in the brain, he wondered?
One theory he’s exploring for removing biofilms involves “debulking” the biofilm slime that shelters bacteria. Since bacterial cells inside biofilms are most impervious to antibiotics when they’re metabolically dormant, or “starving,” he’s looking at ways to “feed” the cells pharmacologically to make them active again. “If you increase the metabolic rate, they become more susceptible to treatment with antibiotics,” Ehrlich says, quoting a paper he published more than a decade ago. “You could get rid of a lot of the cells and, if you get the infection small enough, the host’s immune system might even be able to clear it.”
A Trail of Clues
The idea that Alzheimer’s may be caused by an infection has been catching. In the early ’90s, pathologist and Lyme disease expert Alan MacDonald, currently a research associate at the University of New Haven in Connecticut, originated the theory that Alzheimer’s disease may have a bacterial component. Later, Judith Miklossy, who is director of the International Alzheimer Research Center at the Prevention Alzheimer International Foundation in Switzerland, made a profound observation when she showed that neurosyphilis (a bacterial disease of the brain) and Alzheimer’s were essentially identical on a tissue-level basis.
In a subsequent small study, Miklossy and her colleagues found multiple types of corkscrew-shaped bacteria called spirochetes, which are relatives of the syphilis-causing bacterium, in the brains of more than 90 percent of Alzheimer’s disease patients.
One of the bacteria was Borrelia burgdorferi, which causes Lyme disease. Others were periodontal pathogens closely related to syphilis, which is known to cause amyloid deposition, inflammation and, eventually, dementia similar to Alzheimer’s. Studies by other researchers have made similar connections between bacterial or viral infection and dementia.
The unique pattern of the disease’s progression also hints at an infectious origin.
Ehrlich points out that when the brains of people afflicted with Alzheimer’s are autopsied, it’s possible to see damage to the brain spreading out radially in many directions from the hippocampus, where the nerve cells from the olfactory system terminate. Ehrlich believes that this is because infections are crossing into the brain from the olfactory epithelia.
In fact, one of the first signs that a person will develop dementia is the loss of their sense of smell, he says.
“Our brains are protected by a unique capillary architecture called the blood-brain barrier — but there is one place where the brain is not protected by that barrier and that’s the olfactory epithelium,” he explains. “We know that’s the path herpes simplex virus has used to get to the brain, and we suspect that the pathogens causing Alzheimer’s also use this route.”
Other scientists have also found tantalizing clues suggesting an infection connection. Recently an international group of more than 30 senior scientists and clinicians, including specialists from Oxford, Cambridge, Edinburgh and Manchester universities, authored an editorial in the respected Journal of Alzheimer’s Disease calling upon the scientific community to give the hypothesis credence.
“There is one place where the brain is not protected by the blood-brainbarrier. We suspect the pathogens causing Alzheimer’s use this route.”
They point out that viruses and other microbes are present in the brains of most elderly people. Although usually dormant, reactivation can occur after stress or a treatment from antibiotics. The writers also note that Alzheimer’s has long been known to have a prominent inflammatory component characteristic of infection and, even more interesting, the writers point out that features of Alzheimer’s pathology are transmissible by inoculation of an Alzheimer’s-affected brain to primates and mice.
The authors name the herpes simplex virus — the type that causes cold sores — and Chlamydia pneumoniae as possible culprits. They cite growing evidence in the literature, including more than 100 studies related to the herpes virus alone, that suggest a possible link.
“We propose that further research on the role of infectious agents in [Alzheimer’s disease] causation, including prospective trials of antimicrobial therapy, is now justified,” they wrote.
There was even a documented instance, published in the journal Nature, in which Alzheimer’s-like amyloids were discovered in the autopsied brains of eight adults in the United Kingdom who had died from Creutzfeldt-Jakob Disease. The seeds of Alzheimer’s were believed to have been transmitted through a contaminated medical procedure that all eight patients had received, contradicting the mainstream belief that Alzheimer’s is primarily genetic.
Proving the infection theory — or even disproving it — both carry equal weight, says Drexel’s Saunders. “Following this line of research is so crucial because even if it shows that infection isn’t causative, that’s just as important,” he says. “In our discipline, data talks and what Ehrlich is proposing involves studying thousands of [brain] samples. That will be definitive and help sway what the scientific community thinks.”
Fierce Fight for Dollars
As for why funding agencies and other scientists have been largely unreceptive to the “microbial” Alzheimer’s hypothesis, Ehrlich defers to a quote from German physicist Max Planck: “Science advances one funeral at a time.”
“Scientists are supposed to be the most open-minded people in the world, but many of them are not,” Ehrlich says. He says that many experts in the field conduct a kind of turf war and will actively work to suppress other ideas. “But my question is, if they are such experts, why haven’t they figured out the disease?” he asks.
The Alzheimer’s Association, the largest nonprofit funder of Alzheimer’s research, has said that it is very interested in funding “out-of-the-box” ideas.
“I like to use the hockey analogy — you never know when you’re going to score, so you need as many shots on goal as you can get,” says Keith Fargo, the association’s director of scientific programs and outreach. “We have to have new avenues of research, with people taking a look at this disease from as many different angles as possible.”
But, competition for federal dollars is fierce, Fargo notes.
He says that there was some light at the end of the tunnel in 2011, when President Barack Obama signed into law the National Alzheimer’s Project Act requiring the establishment of a national plan to accelerate the development of treatments for the disease through coordinated research, federal services and funding.
But though the act resulted in a decent bump in research dollars, it still wasn’t quite enough, Fargo says. Currently, the government allocates about $991 million annually for Alzheimer’s and other dementia research, which is a fraction of what is needed to fulfill the national plan’s $2 billion per year goal.
“What that means is that there is very high competition for those research dollars, so difficult decisions have to be made,” says Fargo. “Some avenues that are promising may get overlooked in favor of other avenues that are more developed.”
And while Alzheimer’s disease research is indeed expensive, it’s also quite tricky. The brain is a complex organ made up of billions of cells — “the best estimate is about 80 billion cells with literally trillions of connections — and different cell types,” says Fargo. “It’s a really tough nut to crack.
“In HIV and cancer drug development, there have been some sterling successes,” Drexel’s Saunders says. “Something that was once a death sentence, now isn’t. If cancer shows up in a patient, you can biopsy it and then tailor treatments, but we can’t do that in Alzheimer’s disease, because it’s in your brain. And this is something that probably starts developing in your 40s and manifests when you’re around 75 or 80 — that’s not something we’re used to.”
Ultimately, the scientific community is going to have to marshal a consensus, says Fargo.
“The onus is really on the people who believe in the infection hypothesis to prove it, for lack of a better word,” Fargo says. “Yes, there is a suggestive link, but research has to be done to establish that link.”
