_Steve Wrenn
Wrenn is professor of chemical engineering in the College of Engineering’s Department of Chemical and Biological Engineering.

_Brett Angel
Angel is a cardiologist in the College of Medicine’s Division of Cardiology.

_Aaron Fafarman
Fafarman is an associate professor of chemical engineering in the College of Engineering.
The American Heart Association’s numbers are staggering — 790,000 heart attacks each year; 10 million cardiac-related visits to an emergency room; 17 million deaths from cardiovascular disease worldwide in 2014. The numbers are nearly impossible to grasp…until the moment when a loved one clutches his or her chest and heads to the emergency room.
Even then, the diagnosis can be alarmingly inconclusive.
Doctors must often resort to treatments that are radioactive, invasive and accurate in only about 70 percent of cases. Or they administer stress tests days later, sending a patient home from the emergency room with an uncertain prognosis and deep anxiety about their wellbeing. A spectrum of symptoms may muddy the diagnosis. Disease may hide in the heart’s microvasculature that doctors are unable to detect.
In fact, this may be the most staggering number of all: It is the year 2019, and there is still no broadly reliable medical method to image an incipient heart attack.
But if that’s the Holy Grail of cardiology — and it is — a team of researchers at Drexel may just have entered the shrine.
“This is not pie-in-the-sky. This is working, and the implications are huge. It doesn’t seem far-fetched that we would totally change how chest pain is diagnosed and treated.”
—Steven WrenN
Researchers in the College of Engineering and College of Medicine have developed a groundbreaking invention that can help clinical teams spot a heart at risk before crisis hits.
They created a novel voltage-activated contrast agent that detects heart disease by “lighting up” healthy tissue, while leaving damaged or blocked tissue dark. Called Electrast, the agent responds to the heart’s own electrical activity, rendering it visible with ultrasound when in the presence of the myocardium and providing instant, low-cost, bedside feedback on how efficiently blood is being pumped through the heart.
The journal Applied Acoustics called Electrast the first ultrasound contrast agent to take advantage of the electrical activity of the heart to provide selective activation in the coronary circulation.
Early Validation, Check
Imagine a satellite image of planet Earth at night: Along the coast, major metropolises are lit up like Christmas trees while empty expanses across the heartland are dark voids. That’s essentially what Electrast does for images of the heart: Areas of ample blood flow are brightly illumined, while dark areas indicate a possible stoppage.
Electrast is essentially a contrast agent — that means it is used in conjunction with medical imaging tools like ultrasound to highlight the visibility of bodily fluids and structures. Iodine, for instance, is used in contrast agents injected into patients like a dye to illuminate bodily features within X-ray images.
But Electrast, also delivered by injection, is voltage sensitive so that it reacts to the electric field within the myocardium, the muscular tissue of the heart that keeps blood pumping. It is a phase-change agent that comprises liquid droplets formulated with a novel dual-layer — or nested — architecture. This first layer prevents multiple droplets from sticking to one another; the second layer, the nest, keeps the droplets “silent,” or invisible to ultrasound, until acted on by the electric field to selectively brighten blood flow and illuminate it on an ultrasound.
Electrast has performed superbly in early trials. It has been shown to selectively activate within the heart muscle during small animal studies and in closed-chest infarct validation surgeries in pigs. The latter were carried out by a Drexel team as well as by an independent research group at North American Science Associates Inc. (NAMSA), a contract laboratory in Minnesota.
Under the formal name “Electrophysiologically Activated Intravenous Ultrasound Contrast Agent for Imaging the Heart,” the Electrast project has also received resounding support from the Coulter-Drexel Translational Research Partnership, which shepherds academic research ideas to market that have potential to fulfill unmet or underserved clinical needs. The program has funded Electrast research for three years, the highest level of support.
“We’re hoping to give doctors a better tool so they can answer that question of detection quickly and inexpensively and with minimally invasive technology,” says Steven Wrenn, who is a professor of chemical engineering in the College of Engineering’s Department of Chemical and Biological Engineering and the principal investigator on the Electrast project. “This is not pie-in-the-sky. This is working, and the implications are huge. It doesn’t seem far-fetched that we would totally change how chest pain is diagnosed and treated.”
The medical industry already uses “microbubble” (imagine a soap bubble the size of a red blood cell) ultrasound contrast agents to monitor heart disease. However, these microbubbles have some drawbacks when it comes to evaluating blood flow in the muscle wall. For example, upon entering the body, the microbubbles immediately begin contrasting everywhere so that dead or dying heart tissue is difficult if not impossible to distinguish. In addition, these fragile agents are so abased by ultrasound that their ability to highlight diseased tissue lasts only a couple of minutes.
Electrast surpasses existing agents on both of these scores. Its specificity of activation allows doctors to evaluate blood flow in the essential area — and only in the essential area — with imaging that is of unusually high quality and much longer lasting.
Illuminating Electrast
WHAT IS IT?
Electrast is a voltage-sensitive imaging agent that uses the heart’s electrical activity to selectively contrast high and low regions of blood flow on an ultrasound.
HOW DOES IT WORK?
Electrast becomes visible with ultrasound upon entering the coronary circulation and responding to the heart’s own electrical activity. This provides instant, low-cost, bedside feedback on how efficiently blood is being pumped through the heart.
BENEFITS OF ELECTRAST
_Avoids ionizing radiation.
_Brightens the myocardium yet leaves the ventricle dark.
_Works within minutes and at the bedside.
_Can carry and deliver drugs (e.g., vasodilator) selectively.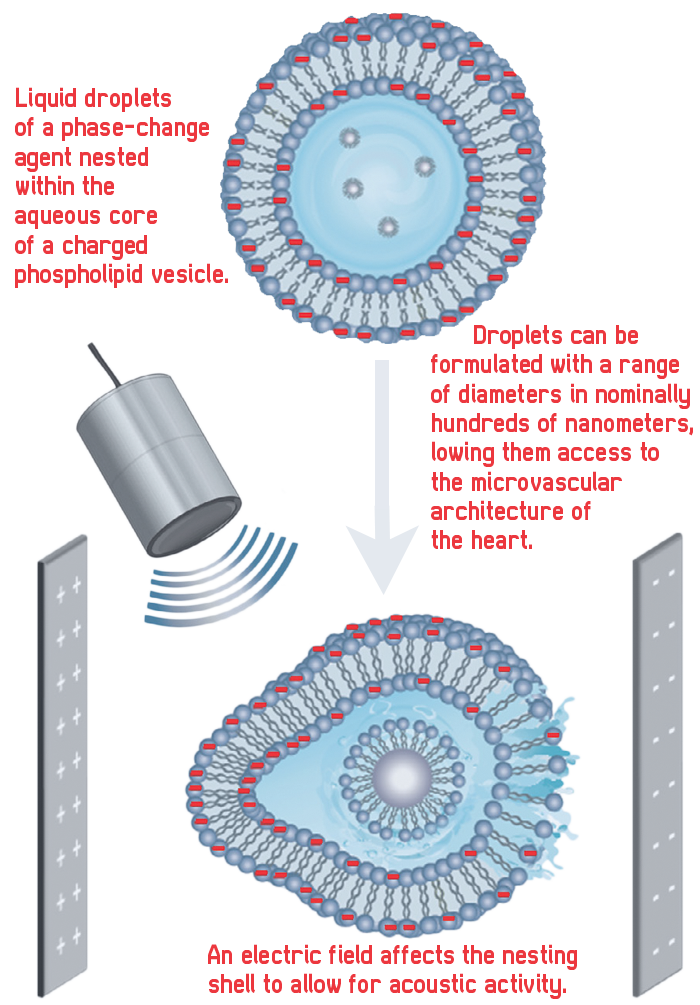
“Where Electrast differs dramatically from traditional microbubbles is that in the blood pool, it travels in a kind of ‘echo stealth mode,’ totally undetectable on ultrasound exam,” says Brett Angel, MD, a cardiologist in the College of Medicine’s Division of Cardiology. “The agent only becomes echogenic and therefore detectable in the small vessels of the heart muscle where it is activated specifically by the electrical activity of viable, living heart muscle cells and the concomitant ultrasound.”
In essence, the Drexel team is coupling the technology of nested droplets with the electrical field of the heart and the acoustic signal of the ultrasound to highlight the myocardium with greater efficiency than ever before.
“If it’s dark, that’s a blockage — it’s really that simple,” says Michael Cimorelli, a graduate student who is pursuing his PhD under the mentorship of Wrenn. “That’s basically what we’ve seen.”
The Heart Is a Muscle
To explain the challenge cardiologists have in tracking the onset of heart disease, Angel offers a well-used emergency room expression: time, he says, is muscle.
Heart disease can build slowly over years before symptoms appear. All too often, by the time a patient gets to the hospital, damage to the heart’s muscle has advanced significantly. In addition, some 40 percent of these patients may have blockages that started in the heart’s network of micro-vessels. Current detection technologies target only the larger vessels.
The ability to catch disease in smaller vessels early would be a huge step forward in cardiac care.
“Cardiovascular disease is ultimately a silent disease until someone has an event,” says Angel. “To this date, there’s very little that you can do to really try and screen for that in terms of imaging the actual blood flow through the heart muscle without exposing a patient to some degree of risk — and without exposing the patient to some degree of ambiguity in the results. There’s a large false positive rate and false negative rate.”
Unlike conventional microbubbles now in use, Electrast droplets can be formulated with a range of diameters in nominally hundreds of nanometers, giving them unprecedented access to the microvascular architecture of the heart.
Wrenn and Angel describe Electrast’s utility as analogous to “silencing the noise in the room,” so that doctors can hear that one voice in the back corner that has something important to say about heart health. The medical industry has been hammering away at techniques that accomplish this for some 30 years with only modest success, which is why Electrast could be a welcome disruption in the medical field.
“This ability to really discern different levels of tissue perfusion, viability and compromise by latching onto the most unique aspect of living cardiac muscle is what makes Electrast such a valuable and disruptive technology,” says Angel.
“On top of that,” says Wrenn, “what we’re putting in — Electrast — is made of ingredients that are fully biocompatible.”
Nesting Instinct
When Wrenn first came to Drexel in the early 2000s, he was working on biological colloids and membranes. He developed an interest in how these structures interacted with ultrasound and how microbubbles could be tethered to the outside of liposomes for controlled drug release within the body. He then had the idea to put the bubble — plus a drug — inside a liposome for a unique, dual-shell architecture now referred to as “the (Wrenn’s) nest.” That architecture allowed for a hardy drug delivery system.
But there was no interest commercially. “They said, ‘that’s encapsulation; there’s nothing new here,’” says Wrenn. “But they were missing the fact that I have two coatings — I have a coating on the bubble at the interface between the gas and the water, but then I have a second shell that’s separated from that coating with water in between — so I have free-floating, coated bubbles inside of a liposome.”
His “nested” structure meant that he could formulate bubbles that persisted indefinitely, instead of dissolving in a matter of minutes.
Wrenn continued studying nesting properties. He generated some 19 papers over eight years on the technology, unwittingly laying the foundation for Electrast.
Then, three years ago, Wrenn got a phone call from a stranger with a pressing question. That stranger was Angel, calling in his excitement from a CVS parking lot.
“It was June 3 of 2015,” Wrenn says. “Brett called me and I had no idea who he was. He was really excited. He had read some of my papers and wanted to know, could I create an agent that could let us see a heart attack in real time? And the main challenge was, could you make it so that it worked with the electrical activity of the heart?
“So I met with him a few times. I began to read and learn about the heart’s physiology. I spent about six months thinking about it before arriving at an idea I thought could work,”says Wrenn. “Then I shared the idea with Aaron [Fafarman] because of his prior experience working with electric fields, and we sketched the idea out on the whiteboard in my office.”
The idea they sketched out on Wrenn’s whiteboard in smudged black ink was that nanodrops could be nested within a voltage-sensitive bilayer of FDA-approved ingredients to create an innocuous phase-change agent that could be deployed in the region cardiologists image to detect early-stage heart disease.
Research began almost immediately in Wrenn’s campus laboratory, the Wrenn Research Group.
BLACKOUT
Electrast spots an imminent heart attack by distinguishing between blood that is in the wall of the heart muscle from blood that is being pumped through the muscle. Electrast is activated only by blood in the muscle wall as the muscle contracts.
In scientific terms, Electrast is an aqueous emulsion of surfactant-coated liquid perfluorocarbon droplets nested within a negatively charged phospholipid bilayer. What that means is that the nest is “decorated” with a charge to make it interactive with an electric field — in this case, the heart’s electric field. Nest sensitivity to voltage allows acoustic activation at an ultrasound intensity that is otherwise insufficient to cause activation. This keeps the droplets invisible to ultrasound until exposure to the electric field in the myocardium.
Thus, with Electrast, cardiologists get localized, high-resolution imaging exactly where it’s needed.
“The heart attack is happening due to a blockage in the muscle, so you want a way to distinguish the blood that’s in the muscle from the blood that’s being pumped by the muscle to keep the heart going. So, you need to be dark in the chamber, bright in the wall,” says Wrenn. “The heart’s depolarization wave [i.e. what makes the heart contract] is happening roughly once a second — lub-dub, lub-dub. It is confined to the tissue of the heart; it’s like dominoes falling, and that signal cascades down and it makes the muscle contract, and that makes the heart pump.
“So, if you can make an agent that is sensitive to that signal, then only the agent that’s in the muscle will be activated and not the stuff that’s farther away. That’s what we’ve done here.”
Coulter Steps In
The Electrast team formed in earnest in 2016 with the first installment of funding from the Coulter-Drexel Translational Research Partnership. Studies followed at Hahnemann University Hospital, at the University of Pittsburgh, and in Minnesota, and began again at Pitt in late January.
The Coulter-Drexel partnership provides funding to between four and six promising Drexel inventions each year to de-risk them for funding milestones on the path to commercialization. “Because we fund early, we are a concierge service,” says Coulter Program Director Kathie Jordan. “What we do depends on what is needed, whether it’s regulatory consulting or finding a CEO — any number of things that it takes so that the project can hit a commercial market.
“Steve was exactly what we look for in projects,” she says of the Electrast. “He was incredibly engaged and excited in the project. One of the things we look for is the strength of the clinical and engineering collaboration, and it was clear right from the beginning that we had a top-notch team.”
Cimorelli says the project is advancing because it brings the problem-solvers, or engineers, together with the day-to-day practitioners, or doctors and medical staff, for maximum proficiency.
“We have two great doctors who really understand the disease states and two amazing engineers with great technical foundations,” Cimorelli says. “I think that’s why this project has been so successful in just two-and-a-half years.”
Coulter funding follows a six-month proposal process that seeks to gauge market need and commercial viability. Since the inception of the Coulter program at Drexel in 2005, 58 projects have received a total of $9.4 million in support. However, the Electrast project is one of only a handful to receive three years of funding. The Coulter partnership has provided Electrast with about $600,000 to date.
The Electrast team met with the Coulter oversight committee again in December to present a film of the surgery being performed at the lab in Minnesota. In the film, the research group performed a progressive series of animal studies to get a sharper image and demonstrate how effective Electrast is in predicting heart attacks. In one surgery, Electrast was deployed via bolus injection. It immediately brightened the subject’s myocardium, leaving an obvious dark spot where the “heart attack” had been created. The brightness was clear even to an untrained eye.
“What venture capitalists want to see is, can you detect a heart attack? They told us: replicate it in the lab, replicate it in the rat, replicate it in the pig. We’ve done all that now,” says Wrenn.
A Super-Fast Camera
The next step for the Electrast research team is to introduce their phase-change agent at cardiology conclaves around the world and begin discussions with venture capitalists about funding.
They’re also working on a series of video microscropy studies at the University of Pittsburgh. There, one of the world’s fastest cameras (25 MHz, or 25 million pictures per second) will provide the team with a frame-by-frame view of exactly how their phase change agent changes.
“We have two great doctors who really understand the disease states and two amazing engineers with great technical foundations. I think that’s why this project has been so successful in just two-and-a-half years.”
—Michael Cimorelli
Electrast is working, says Cimorelli. But there may be ways to tweak and modify the ultrasound’s impact for maximum clarity. They want to establish a nesting efficiency that will give them the best acoustic signature.
They also want to fully understand how it works scientifically, says Cimorelli. “How does the performance respond to all these changes in the chemistry? And then ultimately, we’ll know what combinations to use under certain circumstances.”
“The camera speed is tied to the speed and frequency of the ultrasound,” explains Wrenn. “Typically, when you’re looking at our samples, you’re seeing some brightness but you’re not seeing what’s providing that brightness. Our agent that’s providing the signal is doing things on a megahertz frequency scale — which means millions of times per second, but you’re not seeing that with your eyes, obviously. This camera at Pitt lets you see the actual oscillations, and then you can play it back frame-by-frame and see what is happening each cycle of ultrasound.”
With Coulter’s guidance, the contrast agent will proceed to commercialization at the same time that its mechanism is being more deeply investigated. As team members explain it, the FDA is less concerned with exactly how the phase change agent behaves than that it won’t cause any harm in the body and that it works better than anything else on the market.
For Cimorelli, it’s another matter. “It bugs me like crazy,” he says. “I’m spending 60 hours a week working on this. I want to figure it out. I think it’s valuable to know before it goes to a commercial step, but that’s me.”
Because Electrast is less expensive, non-invasive, portable and wielded with a minimum of training by medical technicians, Wrenn hopes it could someday be used as an annual screening tool. Instead of waiting for an ER episode, general practitioners might use it in office visits to track how a patient with cardiovascular vulnerability looks from year to year — spotting heart disease at much earlier stages than has ever been possible before.
Waxing poetic about the opportunities the team’s contrast agent could afford, Wrenn concludes, “I foresee a day when Electrast is as familiar as an EKG; when hearts brighten on the edge of darkness; and when patients leave a doctor’s office — amazed and perfused.”