_Gail Rosen
Rosen is a professor in the Department of Electrical and Computer Engineering in the College of Engineering. She heads Drexel's Center for Biological Discovery from Big Data.
It’s no secret that we change as we age — we may shrink, we may need glasses and that spring in our step slowly becomes a creak.
But we’re also changing on a sub-cellular level. Our microbiomes — the microbes that live on and within our body — are aging and changing, too.
Gail Rosen, an associate professor in the Department of Electrical and Computer Engineering, and her student Yemin Lan, a doctoral candidate in the School of Biomedical Engineering, Science and Health Systems (BIOMED), have identified markers in our gut bacteria that give away our age, and in doing so, may show the pathway to explain the mechanisms behind some common age-related maladies.
Rosen and Lan took an electrical engineering approach to the research, with help from Andres Kriete, associate director for graduate studies and academic operations in BIOMED. Their work was published in 2013 in Microbiome.
Elderly_Biomes
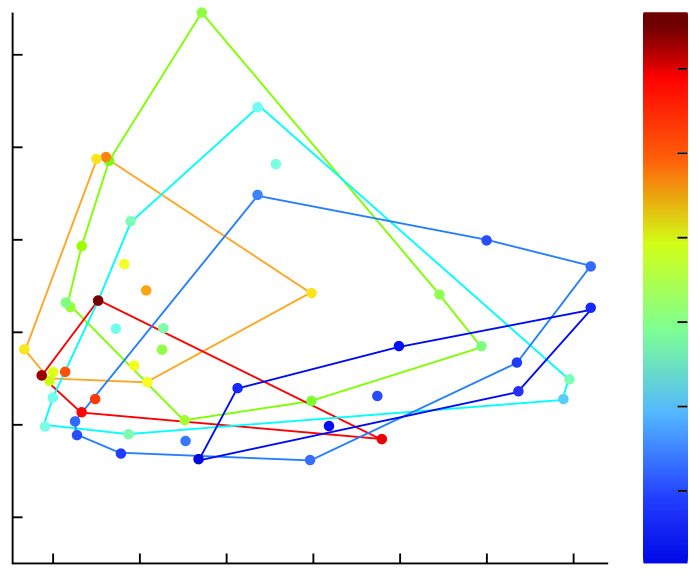
This chart uses the statistical data analysis method known as principal component analysis (PCA) to demonstrate the relationship between the age of individuals in the study sample and certain “selective factors” in their genes associated with aging, such as DNA damage and B12 deficiency. Each dot represents an individual in the sample, marked by a color that corresponds to their age (except for red which indicates 80 years and older). There is a clear differentiation between the oldest and youngest individuals and gradual progression from old to young, indicating that the microbiomes in the elderly behave differently from those of young people. There is also wide overlap among the middle-aged (as is to be expected because individuals age at varying rates).
Specifically, Rosen and Lan looked at the bioflora of patients associated with an inflammatory bowel disease study and differentiated what factors indicated a patient’s age.
They found that the bioflora in older subjects showed a number of factors: reduced activity of reductases, increased DNA damage, compromised stress responses and immune systems. They also found that the bioflora in older subjects synthesized less vitamin B12 than in younger patients.
“It’s well known that the elderly are always deficient in B12 and that they should be taking B12 vitamins,” Rosen says. “This is saying that the microbiome mechanism [for B12 production] may be decreasing as we get older.”
They can now use these selective factors to guess the relative age of an individual’s genetic sample with an “area under the curve” accuracy of 70 percent using the receiver-operating characteristic curve method, which is a way of measuring how well a test correctly classifies information.
Given that people age at different rates, and that our microbiomes vary depending on our environments, 70 percent area under the curve is remarkable. This is the first study of its kind that indicates that aging has a detectable effect on the adult microbiome.
“It’s well known that our eyes get dryer and we don’t generate as much lubrication as we age,” Rosen says. “Could this be affecting our digestive tract and how it hosts the microbiome?” It could be that these age-related changes are involved in a giant feedback loop, and that by understanding the point of age-related breakdown, some age-related body changes can be remedied.